V.I. Nikitenko, DM, Professor, Department of Trauma Surgery, Orthopaedics and Military Surgery; A.A. Stadnikov, DM, Professor, Head of Department of Histology; V.A. Kopylov, Candidate of Medical Science. All at the Orenburg State Medical Academy. Email: bacoren@rambler.ru.
Bacterial translocation from the gastrointestinal tract in healthy and injured rats
Objective: To study the phenomenon of asymptomatic penetration of bacteria from the gastrointestinal tract into various tissues of healthy rats and rats with skin wounds and closed fractures at different doses and with different species of bacteria.
Method: Healthy rats and rats with full-thickness skin wounds and closed femoral fractures were given varying doses of radiolabelled bacteria (S. aureus 209P or B. subtilis 534) per os in doses of up to 1O10 bacteria per 1 kg of body mass. Six hours later, these animals were euthanised and, together with blood smears, tissue samples of liver, spleen, stomach, small and large intestine, lung, hypothalamus, posterior pituitary gland, epinephros and skin and muscle from the wound site were subjected to bacteriological and histological analysis.
Results: In total, 99 rats were used for the experiments. Viable bacteria were found to systemically penetrate into the tissues of the internal organs in 45 healthy animals, 27 rats with incisions and 27 rats with fractures. In healthy animals, viable bacteria mostly accumulated in the stomach wall, the mucous membranes of the small intestine, the mesenteric lymph nodes and in the spleen and liver. Translocation was registered in uninjured animals receiving lx 106 and more bacteria per 1 kg of rat's weight and in injured rats receiving 1 x 104 bacteria and more. Bacteria penetrated the local tissues at both wound sites (skin wounds and fracture sites).
Conclusion: Bacterial translocation is a protective mechanism that gives the immune system constant contact with external antigens, although it can contribute to the spreading of infection in an organism. In healthy animals, small amounts of bacteria penetrate systemically from the tract and small intestine into the tissues of the spleen and liver. During injury, translocated bacteria also appear at the focus of injury.
Conflict of interest: None.
Symptomatic bacterial penetration into the blood from the gastrointestinal tract has been observed following trauma and in patients with various pathological conditions such as tumours, traumatic shock and liver disease. 1-4 It was Berg who first named this phenomenon 'bacterial translocation'.5 In the overwhelming majority of the literature, researchers consider translocation to be a pathological process, developing as a result of stress or systemic diseases such as AIDS.1-4 However, on the basis of our two previous studies,6-7 we consider that translocation of bacteria through the uninjured walls of the gastrointestinal tract may be a powerful protective mechanism. We hold that the symbiosis of tissue and bacteria is possible: bacteria feed on nutrients in the tissues and then have a therapeutic exogenous antibiotic effect. We believe that bacterial translocation from the gastrointestinal tract into tissues is a mechanism for both everyday immunisation and stimulation of immunity in healthy animals.
This hypothesis demanded experimental confirmation, and further study of the phenomenon. Moreover, our literature search had failed to answer the following questions:
Which organs do bacteria translocate to?
Does the phenomenon depend on the dosage of bacteria injected into the gastrointestinal tract per os?
Does bacterial translocation take place in healthy animals?
How does trauma/pathology influence the distribution of bacterial translocation?
Our study set out to answer these questions.
Materials and method
All of our work with laboratory animals complied with the European Convention for the Protection of Vertebrate Animals Used for Experimental and Other Scientific Purposes.8 On 12 April 2005 we gained the approval of our local ethics committee for the following:
The infliction of incisions to the skin and subcutaneous tissues with a scalpel, under anaesthesia
The infliction of closed fractures of the femur with a hammer, under anaesthesia
The introduction of viable Staphylococcus aureus 209P and Bacillus subtilis 534 per os in both healthy and injured rats.
After gaining ethical approval, we selected 99 male Wistar rats weighing 180-230g, all of which were obtained from the nursery at the same time. Rats were given standard nutrition (a variety of foods containing proteins, fats, carbohydrates and vitamins) and water ad libitum. Before experiments, animals were quarantined for 2 weeks. All were healthy and ate well. The animal facility was maintained at a temperature of 20-23°C, with a relative humidity of 43-63%. The animals were kept in portable, 20x50x20cm plywood containers, with a wire mesh top. Bedding was wood sawdust that had not been treated chemically, and each cage contained three animals.
We carried out three different experiments, which are described below.
Translocation threshold To define the minimum number of microorganisms that need to be administered per os before translocation is registered. We took 45 healthy animals and, using a thick, blunt needle, 42 of them were given per os dredges containing different numbers of microorganisms. In short, rats were divided into seven groups of three, with each group receiving one of the following dosages of S. aureus 209P: 1x1O4, 1x1O5, 1x106, 1x107, 1x1O8, 1x109 or 1x1010. An additional seven groups of three rats received one of the following dosages of Bacillus subtilis 534: 1x104, 1x1O5, 1x1O6, 1x1O7, 1x1O8, 1x109 or 1x1010. (The dosage relates to the number of bacteria per kg of body weight.) The number of microbes was calculated per lkg of body weight. As a control, 3 rats who did not receive either S. aureus 209P or B. subtilis 534 were treated similarly to the inoculated rats.
The influence of soft tissue injury on bacterial translocation Under inhalation narcosis, 27 rats were each given one full-thickness lcm wound (by scalpel) through the subcutaneous fat to the broad fascia of the hip. The wound edges were closed in theatre with Steri-Strips (3M Healthcare). Directly after the trauma, rats were divided into eight groups of three animals each, with each group being given one of the following dosages — 1x1O3, 1x1O4, 1x1O5, 1x1O6 — of either S.aureus 209P or B.subtitis 534. (Again, the dosage was of bacteria per lkg of body weight.) As a control, 3 rats who did not receive either S. aureus 209P or B. subtilis 534 were treated similarly to the inoculated rats.
The influence of cortical bone closed fractures on bacterial translocation A further 27 rats were subjected to femoral shaft fractures (using a sterilised hammer), also under inhalation narcosis. Directly following this trauma, animals were divided into the same number of test groups and controls and given the same bacterial dosages as outlined in the second series of experiments above.
Bacterial cultures
In the Orenburg State Medical Academy laboratory, radionuclide [3H]-leucine was added at a dosage of 1MBq to each test tube, which contained 0.75% sterile melted beef-extract agar. Using a sterile loop, nosocomial S. aureus 209P and saprophyte B. subtilis 534 were seeded in glass tubes with slope agar (MICROGEN, Russia). Incubation was maintained at 37°C for 24 hours. (Both S. aureus 209P and B. subtilis 534 were supplied by Tarasevich L.A. Russian State Research Institute of Standardisation and Control of Medical Biological Preparation, Moscow.) Marked microorganisms were then washed out using a sterile solution of NaCl at a dosage of 2cm3 for each test tube. Through adding a 0.9% solution of NaCl to each test tube, dredges with different bacterial concentrations were prepared, each with a volume of 1cm3. We defined the number of microorganisms in each dredge using a spectrophotometer, comparing degrees of transparency against optical turbidity standards (Tarasevich L.A. Russian State Research Institute of standardisation and control of medical biological preparations, Moscow). Using a thick, blunt needle, rats in all the experimental groups were given per os dredges containing marked microorganisms.
Animal sacrifice
Six hours following inoculation, all rats were euthanised with a lethal dose of inhaled halothane. Preliminary studies had suggested that this period is optimal for achieving the greatest bacterial accumulation in the tissues.
Bacteriological studies
Using sterilised instruments and following the thrice-repeated cleansing of skin with a 1% solution of povidone-iodine USP, tissue samples from the spleen, the entire liver, the entire stomach, the small intestine (all parts of the duodenum) and large intestine (all parts) of the inoculated and control animals were taken for bacteriological study. Blood smears from the 'small intestine' were also taken. Tissue samples (1g) were reduced to fragments and pounded in a china vessel with sterilised sand. 10cm3 of 0.9% NaCl was added before transferring to a Petri dish with 1.5% beef-extract agar using a sterilised loop. This was incubated for 24 hours at 37°C. All bacterial species that were found were defined according to Bergey's manual.9 Bacteria were identified by their morphological, tinctorial, cultural and biochemical characteristics, and through the determination of antibiotic resistance.
Histological studies
Together with blood smears, samples of liver, spleen, stomach, small and large intestine, lung, hypothalamus, posterior pituitary gland, epinephros and skin and muscle from the fracture site were processed to single-type for histological handling. Tissue samples were fixed in a 10% neutral buffered formalin. Blood smears for drying were processed with methanol (the slides were expedited in a flask of methanol with a stopper). Later, the fixed organ pieces were dehydrated in alcohol solutions of increasing concentration, and then poured into paraffin-celloidin.
Twenty-one serial sections of 6-8µm were prepared from different parts of each object on rotary microtome. Seven histological slides, with histological cutoffs, were stained with Mayer's hematoxylin and eosin (H&E). The remaining specimens were used for historadiographical purposes, which were carried out in accordance with Ginkin's method.10 Under a red light, the slides were treated with liquid photo emulsion type 'M' (NiiChimPhoto, Moscow) and, following desiccation, were placed in a lightproof container for 4 weeks. They were then processed with amidol developer and sodium hyposulphite (sodium thiosulphate). Radio-autographs were coloured with H&E.
Fourteen sections of each organ were examined for autoradiography. The analysis of bacteria radiolabels number was performed using a measuring grid. The number of radiolabels in a 5x10-4mm2 area were counted in a standard light microscope's field of vision (at 900X magnification). (In each section, 100 squares of the measuring grid in different parts of the histological preparation were analysed, and the results were fixed in conventional units — the average number of radiolabels in one square of the measuring net was 5x10-4mm2).
For electron microscopy, the hypothalamic and posterior pituitary gland tissues were fixed in a 2.5% solution of glutaraldehyde on S-collagen buffer (pH=7.3) and osmium tetroxide. These organs mostly reveal the changes typical for stress.11 Following dehydration in acetone, the pieces were poured into epoxy resin EPON-812. Ultrathin cutoffs, prepared on an LKB-5 ultratome (LKB Instruments, Rockville, MD), were stained with an alcoholic solution of uranyl acetate, followed by aqueous lead citrate. Epon sections were then studied and photographed under an EMB 100AK electron microscope (magnification х6ООО-40000). Ultrastructural analysis was performed in accordance with Hayat's recommendations.13
Results
Translation threshold
Bacteria were not found internally following their per os administration in quantities of 1x104 and 1x105 per 1kg of body weight, but by increasing the dosage to 1x1O6 per 1kg, radiolabels appeared in both the spleen (Fig 1) and liver (Fig 2), but not in the stomach, small intestine and large intestine. In a 5x10-4mm2 area of splenic tissue, there were 0.17 ± 0.03 (mean ± SD) S. aureus radiolabels, and 0.82 ± 0.09 B. subtilis radiolabels. In the liver, there were 0.48 ± 0.05 units of S. aureus and 0.92 ± 0.03 units of B. subtilis. The number of radiolabels observed in the hepatic and splenic tissues did not increase directly in proportion to the dose administered. For instance, increasing the dose of B. subtilis by 10,000 times from 1 x 106/kg (rat) to 1 x 1010/kg, led to a tissue radiolabel increase of just 3.4 times in the spleen and 2.1 times in the liver In the gastric tissues (Fig 3) and small intestine (Fig 4), radiolabelled bacteria were only found after administering doses of 1 x 107/kg and higher. In the large intestine (Fig 5), S. aureus and B. subtilis radiolabels were only registered at doses of 1 x 1010/kg. No radiolabels were found in any of the lung, kidney or skin samples, or blood smears. When compared with the control, histological examination of the small intestinal mucosa of rats that had been administered S. aureus at a dosage of 1 x 1010/kg revealed foci of epithelial desquamation, moderate oedema and mild leucocyte infiltration into the lamina propria mucosa. No pathological changes were observed in the liver, spleen, stomach, large intestine, lungs, epinephros or skin of rats that were administered either bacterial species.
The influence of soft tissue injury on bacterial translation
These results show the influence of incisions on the dynamics of bacterial translocation. When S. aureus and B. subtilis were administered at doses of 1 x 104 kg, their radiolabels were found in the hepatic and splenic tissues, and at the wound site (Fig 6). When S. aureus was administered at a dose of 1 x 104/kg, a mean of 0.76 ± 0.19 radiolabels were found in the liver, 0.19 ± 0.08 in the spleen and 0.95 ± 0.08 in the wound site tissues, all within a 5 x 10-4mm2 area. Following the administration of B. subtilis at the same concentration, an average 0.81 ± 0.21 radiolabels were counted in the liver, 0.54 ± 0.13 in the spleen, and 1.53 ± 0.07 in the wound site tissues. Increasing the bacterial dosage by 100 times (from 1 x 104/kg to 1 x 106/kg) did lead to a rise in the number of radiolabels found in these tissues, but only by 2.5 times.
Translocation to the wound site tissues did take place at dosages of 1 x 104/kg and above, and was more pronounced with B. subtilis than S. aureus. After administering B. subtilis at a dose of 106/kg, for instance, the number of radiolabels in the wound site tissues (skin and subcutaneous fat) was 2.23 ± 0.17; in the same tissues, animals that had received S. aureus (at the same dosage) had 2.8 times fewer radiolabels (0.80 ±0.03).
The influence of cortical bone closed fractures on bacterial translocation
Radiolabelled bacteria were found in liver, spleen and fracture site tissues (Figs 7 and 8) following the administration of bacteria at doses of 1 x 104/kg and higher. Administering S. aureus at a dose of 1 x 104/kg resulted in a mean 1.2 ± 0.09 radiolabels in the liver, 0.34 ± 0.08 in the spleen, 0.35 ± 0.04 in the stomach wall and 0.41 ± 0.05 in the fracture site tissues (all within a 5 x 10-4mm2 area). A 100-fold increase in bacterial dose (to 1 x 106/kg) was associated with an increase in the number of radiolabels observed in the internal organs. For instance, following the administration of S. aureus at a dose of 1 x 104/kg, 0.35 ± 0.04 radiolabels were seen in stomach tissue; after administering the same bacteria at a dose of 1 x 106/kg, 0.72 ± 0.07 radioiabels were seen.
However, the same was not observed in fracture site tissue samples: the number of S. aureus radiolabels did not appear to rise with the same per os dosage increase: 0.41 ± 0.05 for 1 x 104/kg and 0.47 + 0.05 for 1 x 106/kg. With B. subtilis, translocation to the fracture site tissues was pronounced, and more radiolabels were seen with this same dosage increase (rising from 1.9 ± 0.05 to 2.41 ± 0.12).
Thus, the bacterial translocation of B. subtilis and S. aureus from the gastrointestinal tract takes place both in the wound tissue and closed fracture area. Furthermore, the translocation of B. subtilis is more active than that of S. aureus.
Bacterial cultures
Our bacteriological results prove that translocated microorganisms remain viable in the tissues. Culturing tissue samples of rats that had been inoculated with S. aureus revealed growth of the species from 50.0% and 66.7% of the liver and spleen samples, respectively, 41.7% of both the stomach and small intestine tissue samples, 62.5% of the cut wound tissue samples and 70.8% of the fracture site tissue samples. There was no growth in the blood cultures and no growth of either S. aureus or B. subtilis from the stomach, small intestine, liver or splenic tissue samples of any of the control animals.
In the animals inoculated with B. subtilis, viable bacteria were found in 62.5% and 75% of liver and spleen samples, respectively, 50% of the gastrointestinal tract tissues and 87.5% of small intestine homogenate. Viable B. subtilis were also found in 75% of cut wound tissue samples and 56.2% of fracture site tissue samples.
Histological studies
Electron microscopy of tissue samples from rats given S. aureus at doses of 104-108/kg revealed the ultrastructure of terminal axons of neurosecretory cells of the supraoptic and paraventricular nuclei of the hypothalamus to be comparable to those observed in the controls (Fig 8). Examining the perikaryonal cytoplasm of these cells revealed unchanged endoplasmic reticulum tubules and mitochondria, and secretory granules localised to the periphery.
When examining tissue from animals that were administered 104-108 bacteria per kilo, nuclei and nucleoli did not differ from the control parameters, although they increased in size with bacterial doses of 1010/kg. The size of the nuclei of neurosecretory cells increased, on average, by 1.2 times and that of the nucleoli 1.4 times. Morphometric indicators of functionally active neurosecretory cells, the supraoptic and paraventricular nuclei, did not differ significantly in these animals. Analysis of the magnocellular nuclei of the hypothalamus showed balance between the synthesis and release of neurohormones — typical of normal functioning of the hypothalamic-pituitary neurosecretory system.
Histological examination of the adrenal cortex did not reveal any significant differences in the glomerular, fascicular or reticular zones of any of the samples. Corticocyte structure was normal and microvascular vessels had no signs of significant cytological changes.
These findings clearly demonstrate that the oral introduction of S. aureus did not cause any reactive changes in the hypothalamus, neurohypophysis or adrenal glands that were consistent with stress reorganisation.
The translocation of B. subtilis did not cause any structural changes in the wound site tissues in either wounding sample (bone or soft tissue). However, 6 hours after inoculation with S. aureus, the cut wound site tissue had a typical histological picture of alteration and exudation.
In the fracture site tissue of rats receiving S. aureus, some changes did take place, with neutrophilic infiltration of the intramedullary and Haversian canals (by polymorphonuclear leucocytes), and disintegration and cytolysis (disintegration) of tissue at the fracture foci. Where there were soft tissue haemorrhages, cellular infiltration by neutrophils and mononuclear cells was observed. At sites distant from the fracture foci, the bone was histologically normal. In the closed fracture control group, we observed small haemorrhages, dilatation of microvascular vessels and marginal standing of leucocytes. Cellular infiltra¬tion was less pronounced than in inoculated rats.
Discussion
It has been suggested that bacterial translocation only occurs when pathology or trauma affect the mucous membranes of the gastrointestinal tract.13-15 Our results illustrate that bacterial translocation can take place in healthy animals, and that viable bacteria can pass through uninjured mucous membranes of the gastrointestinal tract, particularly those of the small intestine. Our study further shows that in healthy animals there is practically no translocation through mucous membranes of the large intestine. It could be that, due to the relatively short exposure time, the bacteria did not reach the distal part of the gastrointestinal tract. Furthermore, parts of the radiolabelled bacteria may have been absorbed before reaching that far along the gastrointestinal tract.
We consider this relative lack of translocation through the large intestinal wall to be particularly important, as it may help explain the pathogenesis of non-clostridial anaerobic infections, which mainly come from the large intestine.16,17 When the normal composition of the intestinal wall microflora is violated (dysbiosis),18 a large number of microorganisms that are usually found in the large intestine can move to the small intestine,19 from where they might translocate. Intestinal dysbiosis is caused by the prolonged use of antibiotics and some other medicines, such as hormones, stress and systemic diseases such as AIDS.19 According to data that we plan to publish later, the prolonged use of cephalosporins can cause non-clostridial anaerobic infections.
No bacteria were found in any of the blood smears taken during our experiments. According to Smirnov et al.,20 the peak of bacteraemia in healthy animals is between 2-10 minutes post-inoculation, and lasts for half an hour. Our blood smears were taken 6 hours post-inoculation. We believe this is why we did not find viable bacteria in the blood of rats.
Our study found that in healthy white rats given bacterial dredges per os, viable microorganisms accumulate in the spleen and liver. In these tissues, microbial antigens have direct contact with a great number of immune system cells. We suppose that this contact is useful for everyday immunity, and in maintaining the macroorganism's levels of antibodies against the most frequently encountered bacteria.
Prior to this study, there were no data in the literature on a per os-administered bacterial translocation threshold dose. In healthy rats, we found this to be 106/kg, and in injured rats it reduced to 104/kg. Our own, as yet unpublished, data suggest that the threshold of translocation is approximately 10-100 times lower in humans. However, this work is ongoing and will be published separately.
The discovery that increasing the oral dose of bacteria by 10,000 times leads to a less than tenfold increase in the number of bacteria observed in the liver, spleen, stomach, small intestine and wound tissues strongly suggests that translocation is regulated by the macroorganism. Specific regulatory mechanisms are yet to be studied, but we hope to research them in the future.
Trauma was found to influence translocation as injured rats (with incisions or closed fractures) had greater numbers of bacteria in their tissues compared with non-wounded animals. Bacterial species was also found to be an important determinant of the extent of translocation, as illustrated by the greater numbers of B. subtilis in the tissues following inoculation with the same doses as for S. aureus. In both species, at doses of up to 1010/kg, no structural changes were registered in the pituitary-adrenocortical axis system.
We found that, when injured, the character of translocation changes, as bacteria move to the injury site. However, this was not associated with suppuration — there were neither clinical nor morphological signs, and the number of bacteria in situ was a thousand times less than reported under suppuration.21
We believe that translocation likely has both protective and pathological roles. Pathologically, it has the potential to distribute infecting microorganisms systemically and cause damage to previously healthy tissues, yet by presenting environmental antigens to immune cells it can boost immunity. Even S. aureus may be useful in some cases, although its uncontrolled reproduction can cause harm.
As a natural protective mechanism, translocation likely predates cell immunity since it occurs not only in warm-blooded animals but also in bees and plants. It is already being exploited for medical benefit. In Russia, the medicine Sporobacterin (Bacoren, Russia), which contains Bacillus spp. has already been licensed. This is prescribed per os and is used to treat different kinds of surgical infection, including sepsis.
It is not inconceivable that, by harnessing translocation, B. subtilis and other bacteria might be used to manufacture substances with medicinal properties directly at injury sites. Antibiotics, proteolytic enzymes and growth factors are all used in the prophylaxis and treatment of various infections and diseases; if produced in situ, some healthy tissues and organs might be spared. Our team has previously, through the prescription of viable B. subtilis per os, prevented suppuration in shot wounds in rabbits.7 We are amazed at how brilliantly nature solves the difficult problem of how to selectively deliver medicines.
By understanding bacterial translocation, it follows that the tissues of a macroorganism's internal organs are not always sterile. We propose that short-term symbiosis with bacteria is quite possible. This phenomenon offers a macroorganism's immune system direct contact with microbial antigens, which provides everyday immunisation. We hope that, in the future, bacteria might be used to selectively deliver medicines to injured tissues, produc¬ing a medical effect.
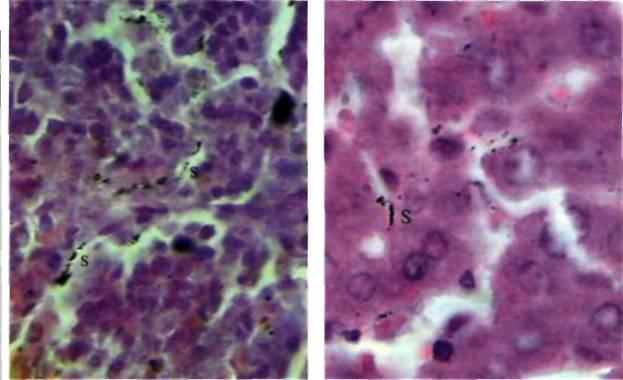
| Fig I. Rat spleen following inoculation with 107 bacteria per 1kg of body mass. Staphylococcus radiolabels are marked 'S'. Magnification: 900X | Fig 2. Peripheral liver lobe following inoculation with 107 bacteria per 1kg of body mass. Staphylococcus radiolabels are marked 'S' Magnification: 900X |
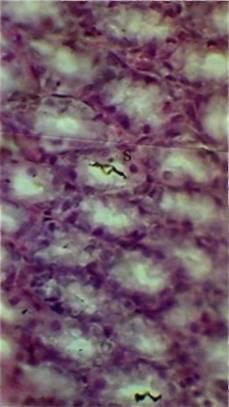
Fig 3. Gastric lining following inoculation with I07 bacteria per 1kg of body mass. Staphylococcus radiolabels are marked 'S' Magnification: 400X
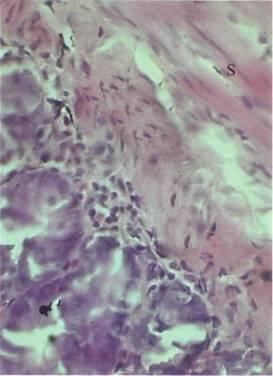
Fig 4. Small intestinal wall following inoculation with 107 bacteria per 1 kg of body mass. Staphylococcus radiolabels are marked 'S' Magnification: 400X
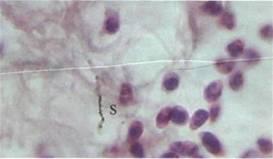
Fig 5. Large intestinal lining following inoculation with 107 bacteria per 1 kg of body mass. Staphylococcus radiolabels are marked 'S'. Magnification: 800X
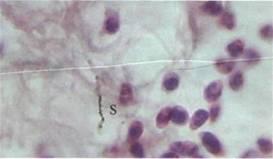
Fig 6.Wound site skin, following inoculation with 104 bacteria per 1 kg of body mass. Staphylococcus radiolabels are marked 'S' Magnification: 900X
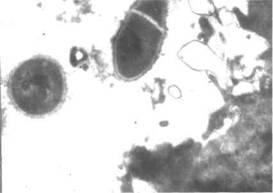
Fig 7. Electron micrograph showing Bacillus subtilis 534 at a closed fracture site following inoculation with 104 bacteria per 1 kg of body mass. Magnification: 30500X
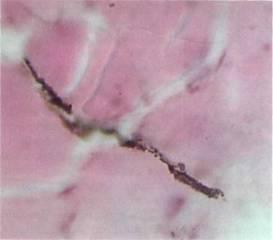
Fig 8. Closed fracture field of femoral diaphysis of a rat following inoculation with 104 bacteria per 1 kg of body mass. Magnification: 900X
References
- 1 Sakamoto, H., Naito, H., Ohta,Y. et. al. Isolation of bacteria from cervical lymph nodes in patients with oral cancer. Arch Oral Biol. 1999; 44: 10, 789-793.
- 2 Demetriades, D., Smith, J.S., Jacobson, L.E.et.al. (1999) Bactericidal/permeability-increasing protein (rBP121) in patients with hemorrhage due to trauma: results of a multicenter phase || clinical trial. rBP121 Acute Hemorrhagic Trauma Study Group. J Trauma. 1999; 46: 667-676.
- 3 Almedia, J., Galhenage, S., Yu, J. et. al. Gut flora and bacterial translocation in chronic liver disease. World J Gastroenterol. 2006; 12: 10, 1493-1502.
- 4 MacFie, J., Reddy, B.S., Gatt, M.et. al. Bacterial translocation studied in 927 patients over 13 years. Br J Surg.2006; 93:1, 87-93.
- 5 Berg, R.D., Garlington, A.W. Translocation of certain indigenous bacteria from the gastrointestinal tract to the mesenteric lymph nodes and other organs in a gnotobiotic mouse model. Infect Immune. 1979; 23:2, 403-411.
- 6 Nikitenko, V.I. Can translocated bacteria reduce wound infection? J Wound Care. 2007; 16: 4, 151-153.
- 7 Nikitenko, V.I. Infection prophylaxis of gunshot wounds using probiotics. J Wound Care. 2004; 13: 9, 363-366.
- 8 European Convention for the Protection Vertebrate Animals used for Experimental and Other Scientific Purposes. Council of Europe , Strasbourg, 1986.
- 9 Holt, J.B. Bergey's Manual of Determinative Bacteriology (9th edn). Williams and Wilkins, 1994.
- 10 Ginkin, L.N. The usage of the radionuclides in histology. The radioactive indicators in histology. LEM AMN USSR 1959; 5-33.
- 11 Stadnikov, A.A. The role of hypothalamic neuropeptides during interactions pro- and eukaryotes (structural-functional aspects). Ekaterinburg. UrBranch RAS 2001; page 244).
- 12 Hayat M. Principles and Techniques of Electron Microscopy (3rd edn). Macmillan. 1989.
- 13 Ferrier. L. Mazelin. L. Cenac, N. et al. Stress-Induced disruption of colonic epithelial barrier: role of interferon-gamma and myosin light chain kinase in mice. Gastroenterology. 2003; 125:3.795-804.
- 14 Sakamoto, K.. Mori, Y., Takagi, H. et al. Translocation of Salmonella typhimurium in rats on total parenteral nutrition correlates with changes in intestinal morphology and mucus gel. Nutrition. 2004; 20:4.372-376.
- 15 Woosley, K.P. The problem of gastric atony. Clin Tech Small Anim Pract. 2004; 19: 1, 43-48.
- 16 Brook, I. Importance of encapsulated anaerobic bacteria in mixed infection. Arch Intern Med 1984; 144: 4, 701-701
- 17 Goldstein, E.J. Anaerobic bacteremia. Clin Infect Dis I996; 23 (Suppl). S97-101.
- 18 Hawrelak, JA., Myers, S.R. The causes of intestinal dysbiosis: a review. Altern Med Rev. 2004; 9:2. 180-197.
- 19 Shenderov, B.A. Medical microbial ecology and functional nutrition. V. II: Socially-ecological and clinical consequences of a disbalance of microbic ecology of the person and animals. Grant, 1998.
- 20 Smirnov, V.V., Reznik, S.R., Sorokulova, I.B., Vunitskaya, V.A. [Mechanisms of the occurrence of symptom-free bacteremia). [Russian]. Mikrobiol Zh. 1988; 50:5, 56-59.
- 21 Kusin, M.I., Kostichonok, V.M. [Wounds and wound infection]. [Russian]. Moscow, Medicine, 1981.

.jpg)